

 |
A control electronics B hydrogen/oxygen producer C battery
|
 |
Control electronics and hydrogen/oxygen (H/O) generator |
 |
Hydrogen oxygen generator in
the detail
(H/O generator) |
 |
Current supply for
electrolysis in the detail
8 x 1,2 V of accumulators (= 9,6
V) |
 |
Battery luggage and
introduction carburetor in the detail
|
 |
Hydrogen/oxygen producer from
above
|
 |
Fill hydrogen/oxygen of the producer |
 |
From left to the
right: Transformer Circuit analyzer Frequency generator Electrolysis tub |
 |
In the detail
|
 |
Electrode in the detail (in the rasenmaeher one uses) |
 |
Experimental setup (1) to the
high frequency electrolysis
Time: x+0 Experimental
setup: Cathode: Copper with silver coat
(galvanic), Cylinder: Standard art off cylinder approx.
90 x 200 mm. |
 |
1. Test launch
Time: x+0.3
minutes System is filled. |
 |
2. Attempt runs
Time: x+1 minutes
Increased H/O splitting off. |
 |
3. Attempt runs
Time: x+3
minutes H/O splitting off increases with frequency change. Frequency in follow further increased. |
 |
4. Attempt runs
Time: x+4
minutes Frequency is continued to increase to 700Hz. H/O production increases superproportionally. |
 |
5. Attempt runs
Time: x+6
minutes H/O production achieves maximum with 923Hz. |
 |
6. Attempt runs
Time: x+7
minutes H/O production becomes smaller with rising frequency. Frequency is gone back gradually on 923Hz. |
 |
7. Attempt end
Time: x+9
minutes End of the experiment. The frequency was gone back gradually again on 923Hz and held for 32 seconds 923Hz. |
| Summary | ||
 |
 |
 |
| x+0 minutes
Water(Mix) volume of 1,2 litres
|
x+6 minutes
Current supply was stored at
short notice. |
X+9 minutes End of the experiment. Became by 1.2 litres water 0,5 litres during the experiment split up into hydrogen and oxygen. |
| By the experimental setup 0,5
litres became water within 9 minutes with changing frequency split up into hydrogen and oxygen. With constant frequency of 923Hz, a voltage of 4.8 V and a current from 300mA a fragmentation becomes from greater 1 litre/minute achieves. This corresponds to regular grade fuel in for instance a calorific value of 0.7 litres. The energy source used in this experiment consisted of 4 accumulators with in each case 1.2 V and 5000mA capacity. (commercial rechargeable a-cells) From this follows: Constant current from 4 x 1,2 V
of A cells: 4,8 V with 5000mAH 360 minutes of x of 0,7 litres = 252 litres calorific value 1 hour = 42 litres calorific value Differently expressed: With 4 batteries (as for instance
in the flashlight) it is possible to produce per hour hydrogen and oxygen
in a quantity which corresponds to regular grade fuel in approximately 42
litres. Until the batteries are exhausted, they produced for calorific
value, which corresponds at least 252 litres to regular grade
fuel. |
This contains links and some diagrams to help you make your circuit.
Need a software that simulate a circuit before you construct it ? See at http://www.beigebag.com/ad2000.htm
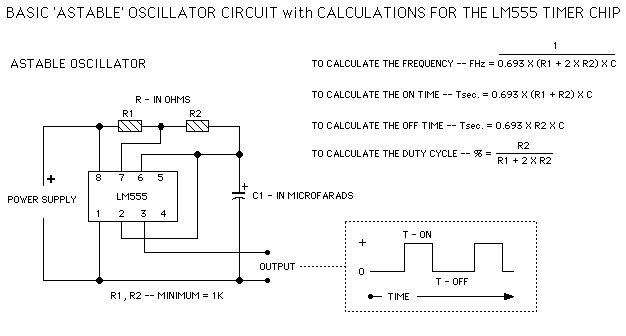
I had some emails assuming that the web owner (me) has performed the above experiment,I have not done so and have merely reproduced the report at this site.
I have had some reports of others conducting this experiment and not getting the results as claimed.
one of these is reproduced as below.
Geoff
I reference to the high frequency electrolysis link on the energy21 terrashare site. I'm a member of an inventor's coop in Northern California and am very interested in researching more efficient means of electrolysis for producing hydrogen on demand for powering vehicles. I have accessed Puharich's patents and white paper and the Xogen patent and other information which you might also find interesting.
I thought of starting with something simple like trying to obtain similar results to your experiment. In trying to replicate or at least approximate the results found in your page on high frequency electrolysis we had very different results.
We utilized an existing power circuit designed by a friend, an electronic engineer. We started with a solution of 10% battery acid, commercially available in the US, It is not clear to us what concentration of sulfuric acid this is, but we added 30ml of this to 270ml of tap water in a plastic beaker. We then used two 24k gold plated copper electrode coils attatched to our power source.
The power source was CURRENT LIMITED to 250mA, the voltage supplied to the circuit was 12V, but as measured at the cell it varied from 1 to 2 volts as the impedance of the cell changed. The cell impedance changed when we varied the frequency of the power source from .6Hz to over 1300HZ. In doing so, we found no change whatsoever in the amount of gas produced as we varied frequency. The only difference we found was that the bubble size changed from large (approx .5cm) to very small.
Because we found that the voltage across the cell varied with frequency we are wondering if perhaps your circuit was voltage requlated with the current varying freely in response to the impedance load of the cell as the frequency varied? If so, this could explain the dramatic difference in gas produced due to current variance.
I am very interested in more efficient electrolysis of water to produce cheap hydrogen and am wondering what we might share to resolve the differences in experimental results?
Thank you for your work and effort put into making so much of this and other information available to people on the internet.
Sincerely, Penn Martin
THIS INFORMATION IS SUPPLIED FOR EDUCATION PURPOSES ONLY AND NO RESPONSIBLY WILL ACCEPTED BY THIS AUTHOR FOR ITS MISUSE.
Have Fun and be care full
Copyright Geoff Egel 2000 Please Share the contents and mention this web site to your contacts and friends.
